High-Resolution fMRI and the Functional Architecture of the Human Medial Temporal Lobe
We are interested in the functional architecture of cognitive functions with a particular focus on memory and the human medial temporal lobe. Characterizing these functional networks in detail in the first place is crucial to understand the effects of Alzheimer’s disease pathology. Thus, we investigate which brain regions and networks are involved in specific memory and other cognitive processes. For example, how our brain can manage to separate very similar experiences without confusing them in memory (Berron et al., JNeurosci, 2016) or how different anatomical pathways are involved in cognitive processing of different material (Berron et al., Neurobiol Aging, 2018).
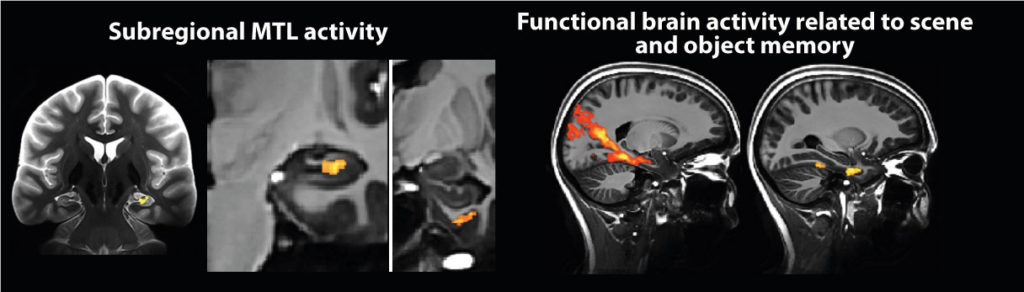
To that end we primarily use high-resolution structural and functional MRI at 3T and 7T to investigate the differential functional connectivity profile as well as the differential involvement of functional brain networks. High-resolution fMRI at 7 Tesla holds great opportunities for research in ageing and neurodegenerative diseases. Thus, we are part of the European Ultrahigh-Field Imaging Network for Neurodegenerative Diseases (EUFIND) where we explore the potential of Ultrahigh-field MRI methods for research in neurodegenerative diseases.
Key publications
Grande X, Sauvage M, Becke A, Düzel E, Berron D. 2022. Transversal functional connectivity and scene-specific processing in the human entorhinal-hippocampal circuitry. eLife.
Berron et al. 2018. Age-related functional changes in domain-specific medial temporal lobe pathways. Neurobiology of Aging.
Berron et al. 2016. Strong Evidence for Pattern Separation in Human Dentate Gyrus. The Journal of Neuroscience.
Koster, R., Chadwick, MJ., Chen, Y., Berron, D., Banino, A., Düzel, E., Hassabis, D., Kumaran, D. 2018. Big-loop recurrence within the hippocampal system supports integration of information across episodes. Neuron.
Maass*, Berron* et al. 2015. Functional subdivisions of the human entorhinal cortex. eLife.
___________________________________________________________________________
Effects of Alzheimer’s Disease Pathology on Medial Temporal and Neocortical Memory Systems
Domain-specific medial temporal lobe pathways support different types of human memory. While brain regions in an anterior-temporal network are more involved in memory for objects, brain regions in a posterior-medial network are preferentially associated with memory for spatial scenes (Berron et al., Neurobiol Aging, 2018). Interestingly, these networks overlap with Alzheimer’s disease pathology in different disease stages. Recently, we could characterize the sequence and dynamics of accumulation of tau pathology across human memory networks using tau PET imaging (Berron et al., Brain, 2021) highlighting that brain areas in the anterior temporal lobe show the earliest signs of tau accumulation.

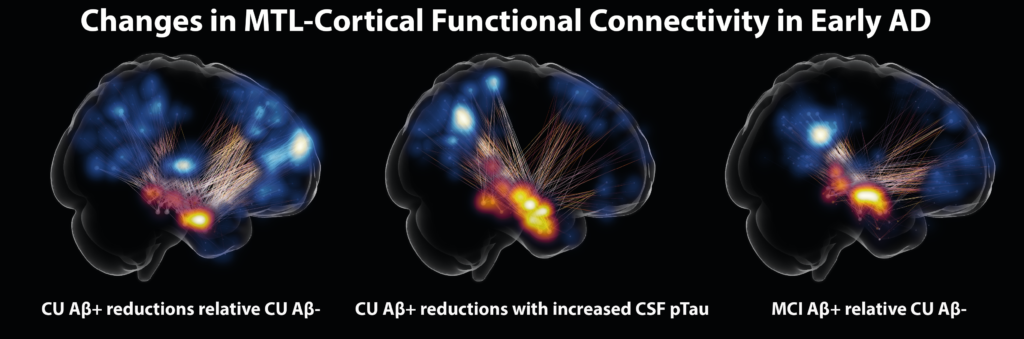
Using functional MRI, biomarkers for Alzheimer’s disease pathology and novel cognitive paradigms, we aim to characterize AD-related changes in functional connectivity as well as in task related activity to define disease stage-specific imaging markers and to understand the origin of early impairment of cognition and memory pathways (Berron et al., JNeurosci, 2019). Recently, we could identify the early changes in MTL-cortical functional connectivity in Alzheimer’s disease, again highlighting the earliest signs of AD-related disconnection in the anterior-temporal network (Berron et al., Brain, 2020).
Key publications
Berron et al. 2021. Early Stages of Tau Pathology and its Associations with Functional Connectivity, Atrophy and Memory. Brain.
Berron et al. 2019. Higher CSF tau levels are related to hippocampal hyperactivity and object mnemonic discrimination in older adults. The Journal of Neuroscience.
Berron et al. 2020. Medial Temporal Lobe Connectivity and its Associations with Cognition in Early Alzheimer’s disease. Brain.
___________________________________________________________________________
Digital cognitive biomarkers to characterize and track cognition in early Alzheimer’s disease
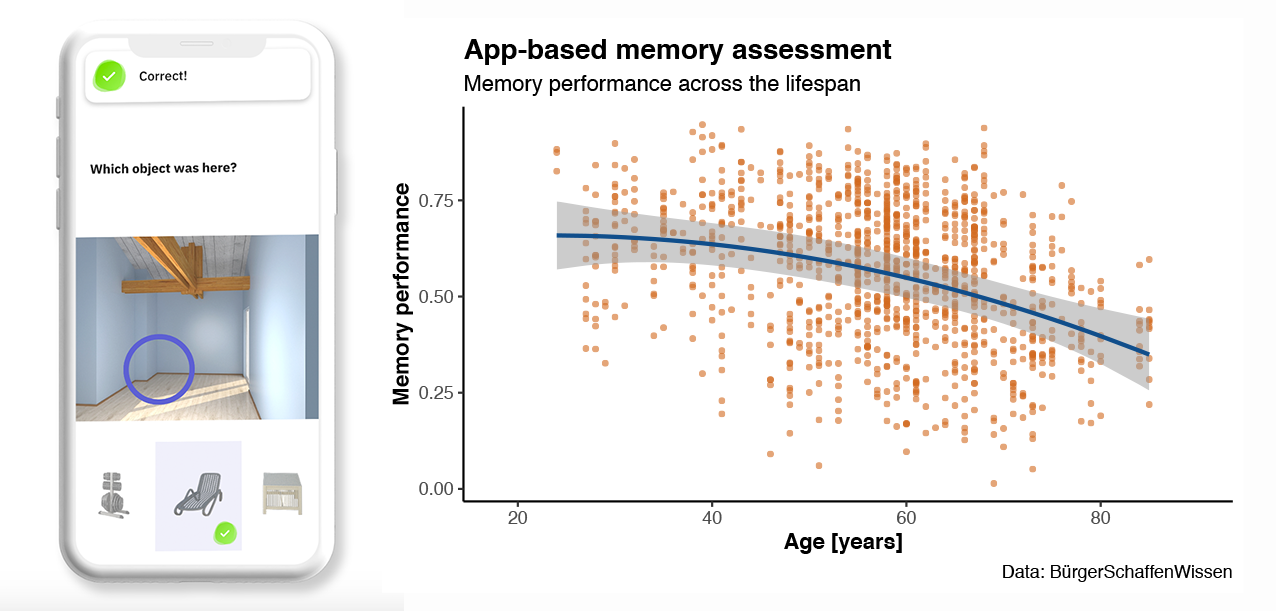 Spatial distribution patterns of Alzheimer’s disease pathology as well as its consequences that can be observed using brain imaging suggest stage- and subtype-specific cognitive impairment in Alzheimer’s disease. Our aim is to understand the earliest cognitive impairment, the sequence of cognitive changes as well as individual cognitive trajectories. To that end, we have developed memory tasks that rely on brain regions that are early affected by Alzheimer’s disease pathology. However, memory performance shows considerable variability across individuals and is influenced by many additional factors that make it difficult to rely on a single test session. Thus, we have translated our memory tasks into smartphone-based assessments which allow for repeated and longitudinal testing of specific memory functions in a remote and unsupervised setting. Recent results from a Citizen Science project within Germany highlight the feasibility and potential of such approaches (in collaboration with university spin-off neotiv that I have co-founded). Using these techniques, we investigate individual trajectories of memory decline in early stages of Alzheimer’s disease using biomarkers for AD pathology based on positron emission tomography (PET), cerebrospinal fluid (CSF) and blood plasma.
Spatial distribution patterns of Alzheimer’s disease pathology as well as its consequences that can be observed using brain imaging suggest stage- and subtype-specific cognitive impairment in Alzheimer’s disease. Our aim is to understand the earliest cognitive impairment, the sequence of cognitive changes as well as individual cognitive trajectories. To that end, we have developed memory tasks that rely on brain regions that are early affected by Alzheimer’s disease pathology. However, memory performance shows considerable variability across individuals and is influenced by many additional factors that make it difficult to rely on a single test session. Thus, we have translated our memory tasks into smartphone-based assessments which allow for repeated and longitudinal testing of specific memory functions in a remote and unsupervised setting. Recent results from a Citizen Science project within Germany highlight the feasibility and potential of such approaches (in collaboration with university spin-off neotiv that I have co-founded). Using these techniques, we investigate individual trajectories of memory decline in early stages of Alzheimer’s disease using biomarkers for AD pathology based on positron emission tomography (PET), cerebrospinal fluid (CSF) and blood plasma.
Key publications
Berron D, Ziegler G, Vieweg P, Billette O, Güsten J, Grande X, Heneka MT, Schneider A, Teipel S, Jessen F, Wagner M, Duzel E. 2022. Feasibility of digital memory assessments in an unsupervised and remote study setting. Frontiers in Digital Health.
Öhman F*, Berron D*, Papp KV, Kern S, Skoog J, Hadarsson Bodin T, Zettergren A, Skoog I, Schöll M. 2022. Unsupervised mobile app-based cognitive testing in a population-based study of older adults born 1944. Frontiers in Digital Health.
Berron D, Glanz W, Billette OV, Grande X, Güsten J, Hempen I, Naveed MH, Butryn M, Spottke A, Buerger K, Perneczky R, Schneider A, Teipel S, Wiltfang J, Wagner M, Jessen F, Düzel E and the DELCODE Consortium. 2021. A Remote Digital Memory Composite to Detect Cognitive Impairment in Memory Clinic Samples in Unsupervised Settings using Mobile Devices. medRxiv.
Berron, D. und Düzel, E.: “Digitales Monitoring von spezifischen kognitiven Beeinträchtigungen in der frühen Alzheimer-Erkrankung“; in Jessen, F., Bug, C. (Hrsg.): “Disease Interception”, eRelation Verlag, Bonn, 2019, S. 79-91 (in german language only, non peer-reviewed)
Düzel E, Thyrian JR, Berron D. 2019. Innovation in der Diagnostik – Mobile Technologien. Der Nervenarzt. (in german language only)
___________________________________________________________________________
Manual Segmentation of Subregions in the Medial Temporal Lobe
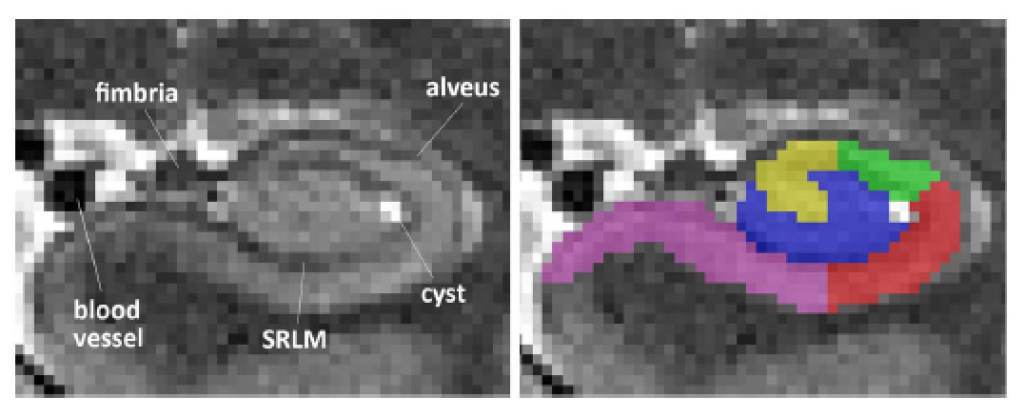
Subregions in the medial temporal lobe are heavily involved in human episodic memory. Recent research showed that subregions are differentially involved in specific memory processes and functions. In order to assess structure-function relationships between the volume and thickness of MTL subregions on one side and specific memory functions on the other, but also to investigate their specific activity profile, regions-of-interest by means of anatomical masks are necessary. Therefore, we developed a manual segmentation protocol for MTL subregions together with colleagues from the DZNE in Magdeburg and collaborators at the University of Pennsylvania and the Allen Institute for Brain Science in Seattle. However, in order to be able to better interpret research on MTL subregions across studies, we are part of the Hippocampal Subfields Group to harmonize segmentation protocols across research labs.
Key publications
Berron*, Vieweg* et al. 2017. A protocol for manual segmentation of medial temporal lobe subregions in 7Tesla MRI. Neuroimage Clinical.
De Flores R*, Berron D* et al. 2019. Characterization of hippocampal subfields using ex vivo MRI and histology data: lessons for in vivo segmentation. Hippocampus.
Wisse LEM, Chételat G, Daugherty A, de Flores R, Mueller S, Stark CEL, Wang L, Yushkevich P, Berron D, Raz N, Bakker A, Olsen R, Carr V. 2020. Hippocampal subfield volumetry from structural isotropic 1mm3 MRI scans: A note of caution. Human Brain Mapping.
Wisse LEM, Daugherty AM, Olsen RK, Berron D et al. 2016. A harmonized segmentation protocol for hippocampal and parahippocampal subregions: Why do we need one and what are the key goals? Hippocampus.